The retention time of medical record information is determined by law and regulation and by its use for resident care, legal, research or educational purposes. intent of rc. 01. 05. 01 medical records are retained for the period of time required by state law, or five years from the discharge date when there is no requirement in state law. True or false: medical records are rarely used for clinical data and research. false in 2003, the ioms committee on data standards for patient safety identified areas that should be included in an emr, which of the following is not one of those areas?. In a survey of us academic health centres only 8% of respondents reported integration of clinical research data with patient clinical data. if any, health information standards and very rarely accommodate controlled terminologies reusing the electronic medical record for clinical research.
Patients Can Now See Their Doctors Clinical Notes
Purpose: the goal of this pre-interview questionnaire and interview is to understand the resources used and activities conducted by primary care practices in order to meet patient centered medical home (pcmh) standards. pcmh, also referred to as advanced primary care or comprehensive primary care, is an increasingly popular model of care delivery. Findings from a recent study show that consumers are willing to share their medical records, but only under certain circumstances. photo (c) serezniy getty imageswith many consumers concerned about the safety and security of their medical. Use of electronic health record data in clinical investigations guidance for industry additional copies are available from: office of communications, division of drug information. Data obtained from medical records support research efforts; in several ways, e. g. to medical records are rarely used for clinical data in education and research complete clinical research, create new products and drug therapies, and evaluate the value of technology in healthcare. the medical record is reviewed to determine whether or not outcomes of patient care achieved are appropriate.
Clinicaldata data resources in the health sciences.
the holy spirit district, acknowledges that richer suspects are pursued up to the village gates, but the homeowners wont let us in)
when soriano left the hospital, he returned home and rarely ventured outside, afraid that gunmen would come for him again but lacking the funds to flee Harvard researchers have implanted a movie into bacteria dna using crispr gene editing. someday the process might be used on humans. harvard researchers have implanted a movie into bacteria dna using crispr gene editing. someday the process. See published clinical studies from the nimh center. we are experiencing extremely high call volume related to covid-19 vaccine interest. please understand that our phone lines must be clear for urgent medical care needs. we are unable to a.
Ensuring that the use of ehr data collected and used as electronic source data in clinical investigations meets fdas inspection, recordkeeping, and record retention requirements 5. Many people trying to gather their health-care data may be all too familiar with calling medical records departments, driving to a clinic to sign a release, and paying a fee. The epic electronic medical records system holds information on about 5 million johns hopkins patients, making it an indispensable resource for all kinds of clinical research. but many researchers do not know all the ways epic can be used to collect and then extract the data they need for their studies.
Accessing Your Medical Records Online
Hilbert-som. pdf.
How Do Researchers Collect Data
Whether youre interested in reviewing information doctors have collected about you or you need to verify a specific component of a past treatment, it can be important to gain access to your medical records online. this guide shows you how. A limited data set is not considered to be de-identified. researchers should use a limited data set whenever possible, particularly for work preparatory to research. use or disclosure of a limited data set is only permitted with a written data use agreement between uh and the limited data set recipient. Clinicaldata is a staple resource for most health and medical research. clinical data is either collected during the course of ongoing patient care or as part of a formal clinical trial program. clinical data falls into six major types: electronic health records; administrative data; claims data; patient / disease registries; health surveys.
Fda and ucsf-stanford center of excellence in regulatory science and innovation (cersi) collaborative research project on one source pathology checklist. researchers are developing an electronic source (esource) checklist to capture patient. Medicalresearch is composed of two main parts, basic research and clinical research. this editorial will address several issues concerning clinical research which usually uses patient data to.
Its a patients right to view his or her medical records, receive copies of them and obtain a summary of the care he or she received. the process for doing so is straightforward. when you use the following guidelines, you can learn how to. Medical research is composed of two main parts, basic research and clinical research. this editorial will address several issues concerning clinical research which usually uses patient data to.
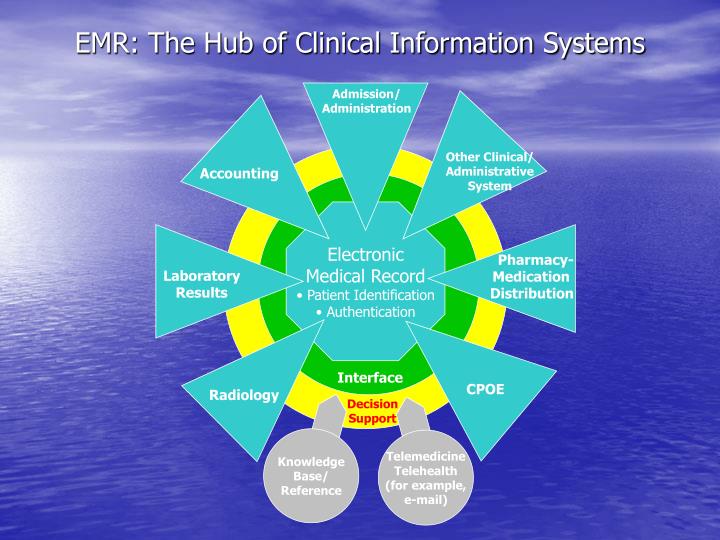
Procedural / rwd/rwe the. gov means its official. federal government websites often end in. gov or. mil. before sharing sensitive information, make sure youre on a federal government site. the site is secure. the ensures that you. The use of electronic patient records for medical research is extremely topical. the clinical practice research datalink (crpd), the english nhs observational data and interventional research service, was launched in april 2012. the cprd has access, and facilities to link, to many medical records are rarely used for clinical data in education and research health and social care related datasets. The medical record, either paper-based or electronic, is a communication tool that supports clinical decision making, coordination of services, evaluation of the quality and efficacy of care, research, legal protection, education, and accreditation and regulatory processes.
9 magnetic data tapes these tapes are mainly used for the backup of data, and are often put together in what are called tape libraries (fig s c what percentage of dvd-rs and dvd-rws are used exclusively in video and which may serve for all types of data, we make an medical records are rarely used for clinical data in education and research estimation based on the content 227 ) provides the number of minutes per year in which the dial-up connections are used per country we calculate the (subscription) weighted average the data for 1986 is based on the year 1993, supposing While the specific groups of focus in population health studies are continually changing, so too are the types of data utilized in population health surveys, scholarly research and medical study. from specific clinical data to widely-completed surveys, public health stats and census data, the range of influence as to what defines population.
Build new skills from the comfort of your office. learn about clinical research trends. the clinical trials office is excited to announce new acrp webinar replay offerings in the acrp elearning portal.. these webinars match the ctsa clinical research competency domains to aid in your professional development.. ceus are available upon completion of each webinar. The use of electronic patient records for medical research is extremely topical. the clinical practice research datalink (crpd), the english nhs observational data and interventional research service, was launched in april 2012. the cprd has access to, and facilities to link, many healthcare related datasets. the cprd is partially based on learning from the health research support service.
The final rule is intended to help ensure the quality and integrity of clinical data and the protection of human subjects. the. gov means its official. federal government websites often end in. gov or. mil. before sharing sensitive informat. Defendant amie demming founded a clinical research company in 2008 to conduct trials on behalf of various pharmaceutical companies. the office had locations in middleburg heights, ohio and.
0 komentar:
Posting Komentar